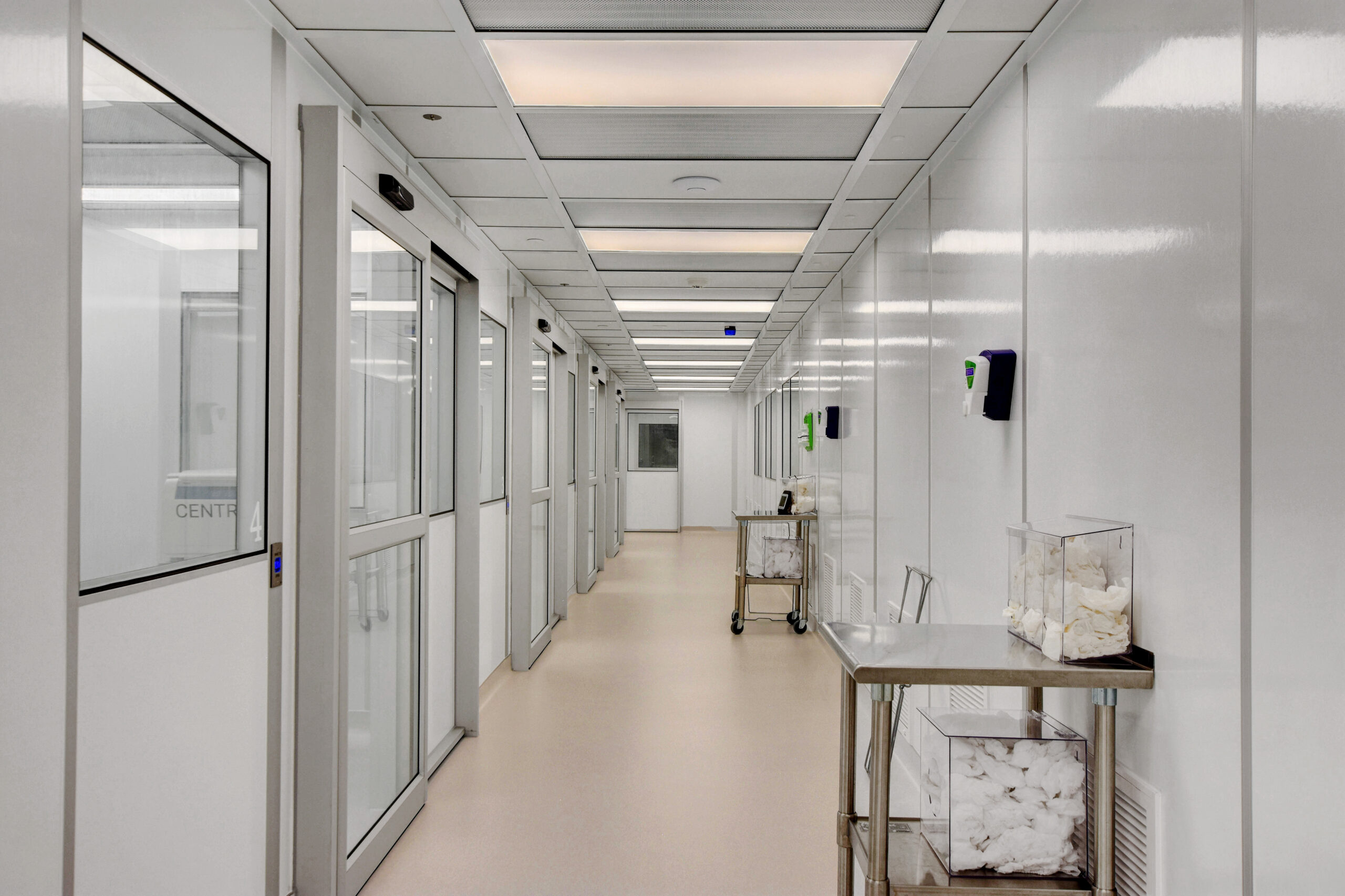
cGMP CLEANROOMS
cGMP stands for Current Good Manufacturing Practices. This standard dictates that certain manufacturers, pharmaceutical packagers, makers of medical devices, and certain food utilize equipment and technologies that are up-to-date with the most current GMP standards. These standards require that the manufacture or packaging of goods adhere to a set of standards that minimizes the risk of contamination, and error to ensure the consumer is receiving the highest quality products.
Pharmaceutical products are subject to Current Good Manufacturing Practices (CGMP). FDA inspectors make sure that a company has the appropriate facilities, equipment, standards, and training to reliably produce high-quality pharmaceuticals. Indeed, we are a top-rated cGMP cleanroom manufacturer and supplier.
cGMP CLEANROOMS
GMP standards give a technically solid approach to consistent and high-quality manufacturing processes for any product seeking market certification. If you’re looking for a cGMP cleanroom, reach out to our specialists today to explore your project and generate a custom quote.
As a best-in-class cleanroom manufacturer, our goal is to help our customers solve problems and deliver an experience that exceeds our customers’ expectations.
To get started on your cGMP cleanroom quote
We will need to gather a little information to ensure a successful project kickoff. Please take a moment to complete the form on this page. Once completed, you can expect a brief call and email from one of our Client Engagement Specialists.
The Client Engagement Specialist will start your quote file and answer any questions you may have regarding your project. During this conversation, please provide Allied Cleanrooms with any drawings or requirements.
Once we have completed a successful kickoff call, our Client Engagement Specialist will connect you with one of our Sales Engineers. Our Sales Engineer will be your direct contact throughout the remainder of your cleanroom needs.
We look forward to working with you to deliver another outstanding Allied Cleanrooms project.